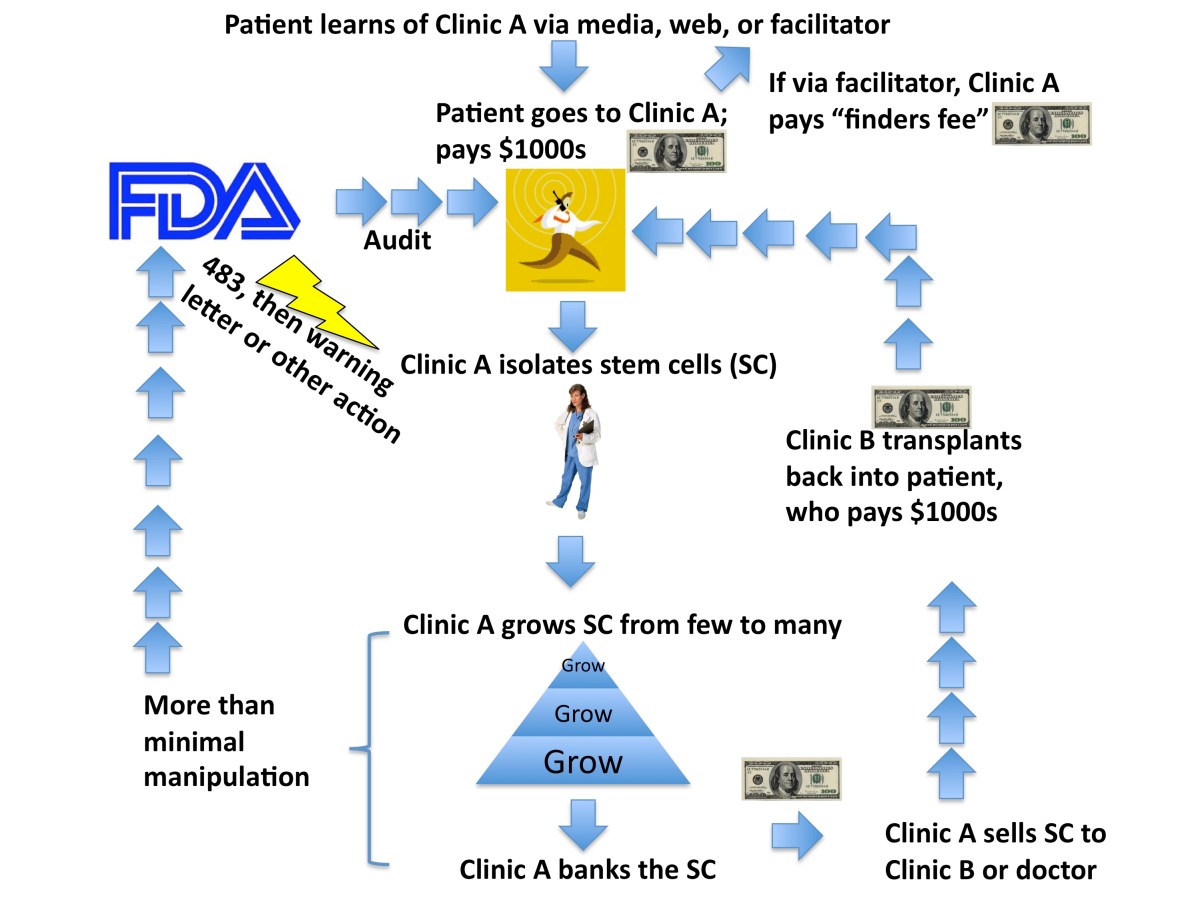
 Adult stem cell clinics are growing fast across the U.S. and the world.Many questions surround them. How do they get patients? How does the money flow? How does the FDA view them? How do all the different parties interact?In science when we study how various proteins or other molecules interact as a system we sometimes call it an “interactome”.I’ve attached a handy-dandy flow chart of my impression of the adult stem cell interactome. A key factor in this system is money.
Adult stem cell clinics are growing fast across the U.S. and the world.Many questions surround them. How do they get patients? How does the money flow? How does the FDA view them? How do all the different parties interact?In science when we study how various proteins or other molecules interact as a system we sometimes call it an “interactome”.I’ve attached a handy-dandy flow chart of my impression of the adult stem cell interactome. A key factor in this system is money.
Of course there are variations on this system. For example, some clinics simply take stem cells and inject them back into patients themselves without handing off to another clinic and may or may not grow them first.
Other clinics do allogeneic transplants from one patient to a different one.
But this interactome largely shows you how adult stem cell clinics work and how the money flows, which for many of the clinics is of course the top priority. Note that the FDA may intercede at any step to “visit” or audit a clinic, potentially leading to actions ranging from nothing to more severe steps such as a warning letter or even involving the FBI.
I’ve highlighted two of the most common triggers for the FDA viewing a stem cell product as “more than minimally manipulated” (you can learn more about that here), but there are many other possible triggers and/or other concerns that the FDA might note.
The total costs typically add up to 10s of thousands per patient.
Good point about costs as they add up.
You also have the model where clinic a> charges initial web consult fee
2-300.00> sales talk etc, more money on office consult sometimes first consult is added in gratis > diagnostics> selective blocks> mri, xray,> pretreatment customized to patient > supplements, (they can choose not to buy or buy elsewhere but get the quality begins here push…we sell the best) infrared, other treatments like IMS, Prolo, Platelet Lysate, PRP, fibrin patches, ETPS nerve blocks etc to diagnose other body parts and best prepare for> stem draw plus>stem cell storage> facility fees ? plus > patient registry 350.00treatment> treatment> usually 3> several thousand I think 7500 each> some do insurance where if treatment doesn’t work there is cost sharing for duplicating treatments. Oh yes then there are fines if you speak about unsuccessful treatment but you are told it is experimental no guarantees…people feel grateful even if they pay money to be diagnosed as poor or fair and think that if they have the treatment it was their choice and their loss. I guess this is considered the practice of medicine?